Properties
Docking into the cavity of Cytochromes or FMO or AOX (Aldehyde oxidase)
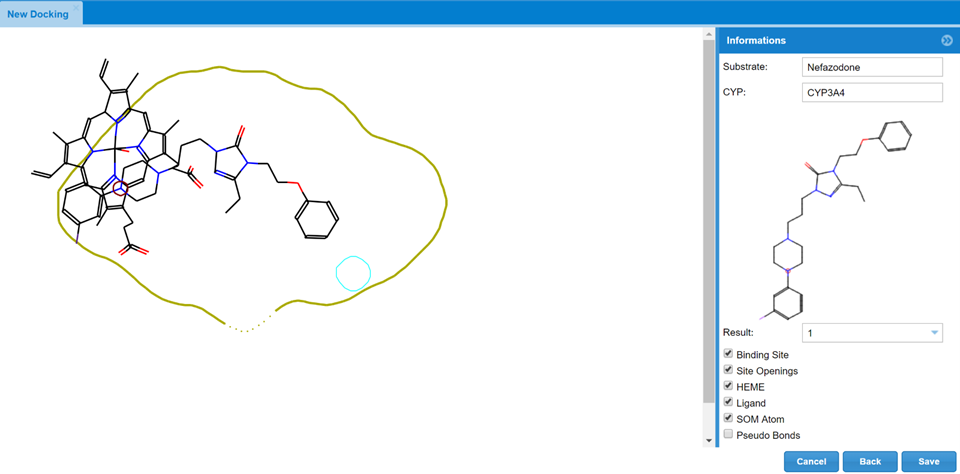
This tool aims to place the parent compound in the cavity of 3A4, 2D6 or 2C9 with the site of metabolism nearby the catalytic point of reaction of the binding site. In the case of the cytochrome P450 family this means to perform a restricted docking where the place where the metabolic reaction occurs (Site of Metabolism) is in the close vicinity of the oxyferryl species. This tool is designed to help the chemists in the understanding of the metabolism of the substrate by showing the potential interactions of the molecule with the different cytochromes provided a site of metabolism.
The user must select one of the metabolites under investigation, the system translates the atom(s) that have been transformed in the parent (SoM) to produce the structure of the metabolite and by selecting the cytochrome the computation will place the substrate with the atom that has suffered the metabolic reaction in the vicinity of the catalytic site of the CYP450, the heme group. This process is done in a 3D mode using the GRID Molecular Interaction fields and the Flap approach. The visualization of the results as done in such a way that the result orientation is reported in the Docking tool that has different layers of information:
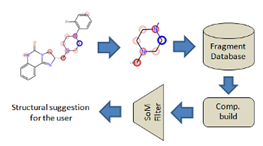
MetaDesign: Suggestion of molecular fragment replacements
This tool is designed to help the user in the design of new molecules having the same boosterism properties than the one under study by replacement of a selected part of the compound.
The workflow starts from a molecule and the selection of a fragment (query) that the user wishes to replace. The query-fragment can be selected manually or automatically based on the Site of Metabolism (SoM) of a detected metabolite. Once the fragment has been selected, it is compared by different techniques to an existing database of fragments, and the ones with the higher bio isosteric index are then processed to build new molecules where the query-fragment is replaced by the solution proposed by the search engine.
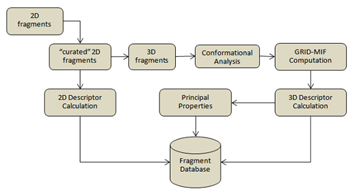
The results proposed by the methodology would depend on how the database of fragments has been built and on the search engine. In the next figure the process to build the database is shown. The starting point is a 2D Sdf file that contains Du to indicate the anchorage point between the fragment and the rest of the molecule. The 2D compound is then processed using the Moka tools (1,2), to find the right ionization state and tautomer at pH 7. The “curated” 2D structures are then processed to compile VS+ (3) like descriptors that describe the Physico-chemical properties of the compounds. The 2D structures are then converted in 3D and a random conformation search is performed. Each conformation is then submitted to the calculation of GRID molecular interaction fields (Hydrogen bond donor, acceptor, positively and negatively charge and hydrophobic interaction) (4). The 3D fields are transformed in a 2D vector that represents the interactions in 3D as a distance-based function (5,6). This set of descriptors is used to determine a principal component space where the query-fragment can be projected. The final database is keeping information about the Physico-chemical properties of the compounds, the distance-based function of the interaction and the principal component scores.
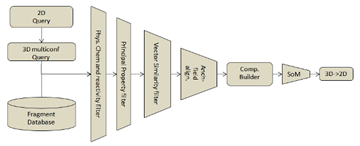
To search the query-fragment into the database it is converted to 3D and a conformation analysis is done, the search in the database is done on each of the conformers that have been found. The search strategy has seven processing steps:
Physico-chemical property filter: The fragments in the database are compared to the query one and only those ones within a certain window in the Physico-chemical parameters are selected. In addition, if the reactivity filtering choice is set ON, only fragments with lower reactivity towards oxidation than the query is selected for the next step.
Principal property filtering. In this step the query-fragment is projected into a principal component space created by the fragments in the database. The fragments that are withing a certain Euclidean distance to the scores of the query one is selected for the next step.
Vector similarity analysis. (2D Similarity Score) The distance-based interaction vectors of the query are compared with the ones in the reduced set of fragments from the database. The ones that pass a certain threshold in the similarity between these fragments are selected for the next step.
The remaining fragments are then used in a Molecular interaction Field alignment in 3D. Each fragment that has been selected is aligned with the query one searching for the maximum overlap of the interaction fields in 3D (3D Similarity Score). The fragments that pass a certain threshold are used in the next step.
The selected fragments are used to build new molecules (the designed compounds) optimizing the 3D geometry of the junction between the fragment and the rest of the molecule.
Finally, the new designed molecules are converted to 2D for the user inspection.
Bibliography
- Extending pKa prediction accuracy: high throughput pKa measurements to understand pKa modulation of new chemical series. Milletti F, Storchi L, Goracci L, Bendels S, Wagner B, Kansy M, Cruciani G. Eur J Med Chem. 2010 Sep;45(9):4270-9.
- Tautomer enumeration and stability prediction for virtual screening on large chemical databases.MillettiF, Storchi L, Sforna G, Cross S, Cruciani G. J Chem Inf Model. 2009 Jan;49(1):68-75.
- BDDCS class prediction for new molecular entities.BroccatelliF, Cruciani G, Benet LZ, Oprea TI. Mol Pharm. 2012 Mar 5;9(3):570-80.
- Molecular fields in drug discovery: getting old or reaching maturity? Cross S, Cruciani G. DrugDiscovToday. 2010 Jan;15(1-2):23-32.
- Anchor-GRIND: filling the gap between standard 3D QSAR and the Grid Independent descriptors. Fontaine F, Pastor M, Zamora I, Sanz F. J Med Chem. 2005 Apr 7;48(7):2687-94.
- SHOP: ScaffoldHOPpingby GRID-based similarity searches. Bergmann R, Linusson A, Zamora I. J Med Chem. 2007 May 31;50(11):2708-17.
